Boron Family
Category : JEE Main & Advanced
Group 13 of long form of periodic table (previously reported as group III A according to Mendeleefs periodic table) includes boron (B) ; aluminium (Al) , gallium (Ga), indium (In) and thallium (Tl) Boron is the first member of group 13 of the periodic table and is the only non-metal of this group. The all other members are metals. The non-metallic nature of boron is due its small size and high ionisation energy. The members of this family are collectively known as boron family and sometimes as aluminium family.
(1) Electronic configuration
| Element | Electronic configuration (\[n{{s}^{2}}\ n{{p}^{1}}\]) |
| \[_{5}B\] | \[[He]\,2{{s}^{2}}2{{p}^{1}}\] |
| \[_{13}Al\] | \[[Ne]\,3{{s}^{2}}3{{p}^{1}}\] |
| \[_{31}Ga\] | \[[Ar]\,3{{d}^{10}}4{{s}^{2}}4{{p}^{1}}\] |
| \[_{49}In\] | \[[Kr]\,4{{d}^{10}}5{{s}^{2}}5{{p}^{1}}\] |
| \[_{81}Tl\] | \[[Xe]\,4{{f}^{14}}5{{d}^{10}}6{{s}^{2}}6{{p}^{1}}\] |
(2) Occurrence : The important of this group elements are given below,
Boron : Borax (Tincal) (Na2B4O7.10H2O), Colemanite (Ca2B6O115H2O)
Boracite (2Mg3B8O15.MgCl2), Boronatro calcite (CaB4O7.NaBO2.8H2 O),
Kernite (Na2B4O7.4H2O), Boric acid (H3BO3)
Aluminium : Corundum (Al2O3), Diaspore (Al2O3.H2O), Bauxite (Al2O3. 2H2O), and Cryolite (Na3AlF6).
Physical properties
(1) A regular increasing trend in density down the group is due to increase in size.
(2) Melting points do not vary regularly and decrease from B to Ga and then increase.
(3) Boron has very high melting point because it exist as giant covalent polymer in both solid and liquid state.
(4) Low melting point of Ga (29.80C) is due to the fact that consists of only Ga2 molecule; it exist as liquid upto 20000C and hence used in high temperature thermometry.
(5) Boiling point of these elements however show a regular decrease down the group.
(6) The abrupt increase in the atomic radius of Al is due to greater screening effect in Al (it has 8 electrons in its penultimate shell) than in B (it has 2 electrons in its penultimate shell)
(7) The atomic radii of group 13 elements are smaller than the corresponding s-block elements. This is due to the fact that when we move along the period, the new incoming electron occupy the same shell whereas the nuclear charge increases regularly showing more effective pull of nucleus towards shell electrons. This ultimately reduces the atomic size.
(8) The atomic radius of Ga is slightly lesser than of Al because in going from Al to Ga, the electrons have already occupied 3d sub shell in Ga. The screening effect of these intervening electrons being poor and has less influence to decrease the effective nuclear charge, therefore the electrons in Ga experience more forces of attractions towards nucleus to result in lower size of Ga than Al
(9) Oxidation state
(i) All exhibit +3 oxidation state and thus complete their octet either by covalent or ionic union.
(ii) Boron being smaller in size cannot lose its valence electrons to form B3+ ion and it usually show +3 covalence. The tendency to show +3 covalence however decreases down the group even Al shows +3 covalence in most of its compounds.
(iii) Lower elements also show +1 ionic state e.g Tl +, Ga+. This is due to inert pair effect. The phenomenon in which outer shell ‘s’ electrons (ns2) penetrate to (n – 1) d-electrons and thus become closer to nucleus and are more effectively pulled the nucleus. This results in less availability of ns2 electrons pair for bonding or ns2 electron pair becomes inert. The inert pair effect begins after n3 4 and increases with increasing value of n.
(iv) The tendency to form M+ ion increases down the gp. Ga+1 < Tl+1
(10) Hydrated ions : All metal ions exist in hydrated state.
(11) Ionisation energy
(i) Inspite of the more charge in nucleus and small size, the first ionisation energies of this group elements are lesser than the corresponding elements of s block. This is due to the fact that removal of electron from a p-orbitals (being far away from nucleus and thus less effectively held than s-orbitals) is relatively easier than s-orbitals.
(ii) The ionisation energy of this group element decrease down the group due to increases in size like other group elements.
(iii) However, ionisation energy of Ga are higher than that of Al because of smaller atomic size of Ga due to less effective shielding of 3d electrons in Ga. Thus valence shell exert more effective nuclear charge in Ga to show higher ionisation energies.
(12) Electropositive character
(i) Electropositive character increases from B to Tl.
(ii) Boron is semi metal, more closer to non-metallic nature whereas rest all members are pure metals.
(iii) Furthermore, these elements are less electropositive than s-block elements because of smaller size and higher ionisation energies.
(13) Oxidation potential
(i) The standard oxidation potentials of these element are quite high and are given below,
B Al Ga In Tl
E0op for M \[\to \] M3++ 3e – +1.66 +0.56 +0.34 +1.26
E0op for M \[\to \] M+ + e – +0.55 – +0.18 +0.34
(ii) However Boron does not form positive ions in aqueous solution and has very low oxidation potential.
(iii) The higher values of standard oxidation potentials are due to higher heats of hydration on account of smaller size of trivalent cations.
(iv) Aluminium is a strong reducing agent and can reduce oxides which are not reduced even by carbon. This is due to lower ionisation energy of aluminium than carbon. The reducing character of these elements is Al > Ga > In > Tl.
(14) Complex formation : On account of their smaller size and more effective nuclear charge as well as vacant orbitals to accept elements, these elements have more tendency to form complexes than-s block elements.
Chemical properties
(1) Hydrides
(i) Elements of group 13 do not react directly with hydrogen but a number of polymeric hydrides are known to exist.
(ii) Boron forms a large no. of volatile covalent hydrides, known as boranes e.g. B2H6,B4H10,B5H11,B6H10 Two series of borones with general formula BnHn+4 and BnHn+6 are more important.
(iii) Boranes are electron deficient compounds. It is important to note that although BX3 are well known, BH3 is not known. This is due of the fact that hydrogen atoms in BH3 have no free electrons to form pp–pp back bonding and thus boron has incomplete octet and hence BH3 molecules dimerise to form B6H6 having covalent and three centre bonds.
(iv) Al forms only one polymeric hydride (AlH3)n commonly known as alane It contains Al…..H……Al bridges.
(v) Al and Ga forms anionic hydrides e.g. LiAlH4 and LiGa H4,
\[4LiH+AlC{{l}_{3}}\xrightarrow{ether}Li[Al{{H}_{4}}]+3LiCl\]
(2) Reactivity towards air
(i) Pure boron is almost unreactive at ordinary temperature. It reacts with air to form B2O3 when heated It does react with water. Al burns in air with evolution of heat give Al2O3.
(ii) Ga and In are not effected by air even when heated whereas Tl is little more reactive and also form an oxide film at surface. In moist air, a layer of Tl (OH) is formed.
(iii) Al decomposes H2O and reacts readily in air at ordinary temperature to form a protective film of its oxides which protects it from further action.
(3) Oxides and hydroxides
(i) The members of boron family form oxide and hydroxides of the general formula M2O3 and M (OH)3 respectively.
(ii) The acidic nature of oxides and hydroxides changes from acidic to basic through amphoteric from B to Tl.
\[\underset{\text{(acidic)}}{\mathop{{{B}_{2}}{{O}_{3}}\,\text{and }B{{(OH)}_{3}}}}\,>\underset{\text{(amphoteric)}}{\mathop{A{{l}_{2}}{{O}_{3}}\text{ and }Al{{(OH)}_{3}}}}\,\]
\[\underset{\text{(amphoteric)}}{\mathop{G{{a}_{2}}{{O}_{3}}\,\text{and }Ga{{(OH)}_{3}}}}\,>\underset{\text{(basic)}}{\mathop{I{{n}_{2}}{{O}_{3}}In\,{{(OH)}_{3}}}}\,>\underset{\text{(strong basic)}}{\mathop{T{{l}_{2}}{{O}_{3}}Tl{{(OH)}_{3}}}}\,\]
B(OH)3 or H3BO3 is weak monobasic Lewis acid.
(iii) Boric acid, B(OH)3 is soluble in water as it accepts lone pair of electron to act as Lewis acid. Rest all hydroxides of group 13 are insoluble in water and form a gelatinous precipitate.
B(OH)3 + H2O \[\to \] B(OH)41– + H+
(iv) Al2O3 being amphoteric dissolves in acid and alkalies both.
Al2O3 + 3H2SO4 \[\to \]Al2 (SO4)3 + 3H2O
\[A{{l}_{2}}{{O}_{3}}+2NaOH\xrightarrow{fuse}\underset{\text{Sodium meta aluminate}}{\mathop{2NaAl{{O}_{3}}}}\,+{{H}_{2}}O\]
(v) One of the crystalline form of alumina (Al2O3) is called corrundum. It is very hard and used as abrasive. It is prepared by heating amorphous form of Al2O3 to 2000 K.
(4) Action of Acids
(i) Boron does not react with non oxidizing acids, however, it dissolves in nitric acid to form boric acids.
(ii) Al, Ga and In dissolve in acids forming their trivalent cations; however, Al and Ga become passive due to the formation of protective film of oxides.
(iii) Thallium dissolves in acids forming univalent cation and becomes passive in HCl due to the formation of water insoluble TICl.
(5) Action of Alkalies
(i) Boron dissolves only in fused alkalies,
\[2B+6NaOH\left( fused \right)\to 2N{{a}_{3}}B{{O}_{3}}+3{{H}_{2}}\]
(ii) Al and Ga dissolves in fused as well as in aqueous alkalies, \[2Al+2\text{ }NaOH+2{{H}_{2}}O\to 2NaAl\text{ }{{O}_{2}}+3{{H}_{2}}\]
(iii) Indium remains unaffected in alkalies even on heating.
(6) Halides
(i) All the group 13 elements from the trihalides, MX3 on directly combining with halogens.
\[M+{{X}_{2}}\to M{{X}_{3}}\]
(ii) All the trihalides of group 13 elements are known except Tl (III) iodide.
(iii) Due to small size and high electronegativity of boron, all boron halides are covalent and Lewis acids. These exist as monomeric molecules having plane triangular geometry (sp2 hybridization).
(iv) All Boron trihalides except BF3 are hydrolysed to boric acid.
\[B{{X}_{3}}+3{{H}_{2}}O\to B{{\left( OH \right)}_{3}}+\text{ }3HX;~~~~~~~~~~~~~~\left[ X=Cl,\text{ }Br,\text{ }I \right]\]
However, BF3 forms as addition product with water,
\[B{{F}_{3}}+{{H}_{2}}O\to {{H}^{+}}[B{{F}_{3}}OH]{{H}_{3}}{{O}^{+}}[B{{F}_{3}}OH]\]
BF3 having less tendency for hydrolysis as well as Lewis acid nature, is extensively used as a catalyst in organic reactions e.g. Friedel- Crafts reaction.
(v) Boron atom, in BX3, has six electrons in the outermost orbit and thus it can accept a pair of electrons form a donor molecule like NH3 to complete its octet. Hence boron halides act as very efficient Lewis acids. The relative Lewis acid character of boron trihalides is found to obey the order ; BI3 > BBr3 > BCl3 > BF3.
However, the above order is just the reverse of normally expected order on the basis relative electronegativities of the halogens. Fluorine, being the most electronegative, should create the greatest electron deficiency on boron and thus B in BF3 should accept electron pair from a donor very rapidly than in other boron trihalides. But this is not true.
This anomalous behaviour has been explained on the basis of the relative tendency of the halogen atom to back-donate its unutilised electrons to the vacant p orbitals of boron atom. In boron trifluoride, each fluorine has completely filled unutilised 2p orbitals while boron has a vacant 2p orbital. Now since both of these orbitals belong to same energy level (2p) they can overlap effectively as a result of which fluorine electrons are transferred into the vacant 2p orbital of boron resulting in the formation of an additional pp–pp bond. This type of bond formation is known as back bonding or back donation. Thus the B- F bond has some double bond character. Back bonding may take place between boron and of the three fluorine atoms and thus boron trifluoride is regarded as a resonance hybrid of some structures.
Resonance in boron trifluoride is also evidenced by the fact that the three boron-fluorine bonds are indentical and are shorter than the usual single boron-fluorine bond As a result of back bonding, the electron deficiency of boron is reduced and hence Lewis acid nature is decreased. The tendency for the formation of back bonding (pp- pp bond) is maximum in BF3 and decreases very rapidly from BF3 to BI3 This is probably due to the fact that overlapping of the vacant 2p orbitals of boron cannot take place easily with the p-orbitals of high energy levels (3p in Cl, 4p in Br and 5p in iodine). Thus BI3Br3 and BCl3 are stronger Lewis acids than the BF3.
(vi) Lewis acid character of halides of the group 13 elements decreases in the order, B > Al > Ga > In.
(vii) Boron halides form complex halides of the type, [BF4–], in which boron atom extends its coordination number to four by utilising empty p-orbital. It cannot extend its coordination number beyond four due to non availability of d-orbitals. However, the other trihalides of this group form complex halides of the type (AlF6)3–, (GaCl6)3– and (InCl6)3–, etc where the central atom extends its coordination number to 6 by the use of d-orbitals.
(viii) The fluorides of Al, Ga In and Tl are ionic and have high melting points. The high melting points of metal fluorides can be explained on the basis that their cations are sufficiently large and have vacant d-orbitals for attaining a coordination number of six towards the relatively small fluorine atom.
(ix) Other halides of Al, Ga, In and Tl are largely covalent in anhydrous state and possess low melting point. These halides do not show backbonding because of increases in the size of the element. However, the make use of vacant p-orbitals by co-ordinate bond i.e. metal atoms complete their octet by forming dimers. Thus aluminium chloride, aluminium bromide and indium iodide exist as dimers, both in the vapour state and in non-polar solvents.
The dimer structure for Al2Cl6 is evidenced by the following facts,
(a) Vapour density of aluminium chloride measured at 4000C corresponds to the formula Al2Cl6.
(b) Bond distance between aluminium chlorine bond forming bridge is greater (2.21 Å) than the distance between aluminum-chlorine bond present in the end (2.06 Å). The dimeric structure disappears when the halides are dissolved in water This is due to high heat of hydration which split the dimeric structure into [M(H2O)6]3+ and 3X– ions and the solution becomes good conductor of electricity.
\[A{{l}_{2}}C{{l}_{6}}+\text{ }2{{H}_{2}}O\to 2{{\left[ Al{{\left( {{H}_{2}}O \right)}_{6}} \right]}^{3+}}+6C{{l}^{}};\] Therefore Al2Cl6 is ionic in water.
The dimeric structure may also split by reaction with donor molecules e.g. R3N. This is due to the formation of complexes of the type R3NAlCl3 The dimeric structure of Al2Cl6 exist in vapour state below 473K and at higher temperature it dissociates to trigonal planar AlCl3 molecule.
Boron halides do not exist as dimer due to small size of boron atom which makes it unable to co-ordinate four large-sized halide ions.
(x) BF3 and AlCl3 acts as catalyst and Lewis acid in many of the industrial process.
Anomalous Behaviour of Boron
Like Li and Be, Boron – the first member of group 13 also shows anomalous behaviour due to extremely low size and high nuclear charge/size ratio, high electronegativity and non-availability of d electrons. The main point of differences are,
(1) Boron is a typical non- metal whereas other members are metals.
(2) Boron is a bad conductor of electricity whereas other metals are good conductors.
(3) Boron shows allotropy and exists in two forms – crystalline and amorphous. Aluminium is a soft metal and does not exist in different forms.
(4) Like other non-metals, the melting point and boiling point of boron are much higher than those of other elements of group 13.
(5) Boron forms only covalent compounds whereas aluminium and other elements of group 13 form even some ionic compounds.
(6) The hydroxides and oxides of boron are acidic in nature whereas those of others are amphoteric and basic.
(7) The trihalides of boron (BX3) exist as monomers On the other hand, aluminium halides exist as dimers (Al2X6).
(8) The hydrides of boron i.e. boranes are quite stable while those of aluminium are unstable.
(9) Dilute acids have no action on boron Others liberate H2 from them.
(10) Borates are more stable than aluminates.
(11) Boron exhibit maximum covalency of four e.g., \[BH_{4}^{-}\] ion while other members exhibit a maximum covalency of six e.g., \[{{[Al{{\left( OH \right)}_{6}}]}^{3-}}\].
(12) Boron does not decompose steam while other members do so.
(13) Boron combines with metals to give borides e.g. Mg3B2. Other members form simply alloys.
(14) Concentrated nitric acid oxidises boron to boric acid but no such action is noticed other group members.
\[B+3HN{{O}_{3}}\to {{H}_{3}}B{{O}_{3}}+\text{ }3N{{O}_{2}}\]
Diagonal relationship between Boron and Silicon
Due to its small size and similar charge/mass ratio, boron differs from other group 13 members, but it resembles closely with silicon, the second element of group 14 to exhibit diagonal relationship. Some important similarities between boron and silicon are given below,
(1) Both boron and silicon are typical non-metals, having high m.pt. b.pt nearly same densities (B = 2.35 gml–1 S = 2.34 g//ml). low atomic volumes and bad conductor of current. However both are used as semiconductors.
(2) Both of them do not form cation and form only covalent compounds.
(3) Both exists in amorphous and crystalline state and exhibit allotropy.
(4) Both possess closer electronegativity values (B = 2.0; Si = 1.8).
(5) Both form numerous volatile hydrides which spontaneously catch fire on exposure to air and are easily hydrolysed.
(6) The chlorides of both are liquid, fume in most air and readily hydrolysed by water.
\[BC{{l}_{3}}+\text{ }3{{H}_{2}}O\to B{{\left( OH \right)}_{3}}+3HCl~\]
\[SiC{{l}_{4}}+{{H}_{2}}O\,\to Si{{\left( OH \right)}_{4}}+\text{ }4HCl\]
(7) Both form weak acids like \[{{H}_{3}}B{{O}_{3}}\] and \[{{H}_{2}}Si{{O}_{3}}\].
(8) Both form binary compounds with several metals to give borides and silicide. These borides and silicide react with \[{{H}_{3}}P{{O}_{4}}\] to give mixture of boranes and silanes.
\[\begin{array}{*{35}{l}}3Mg+2B\to M{{g}_{3}}{{B}_{2}};M{{g}_{3}}{{B}_{2}}+{{H}_{3}}P{{O}_{4}}\to \text{Mixture of boranes} \\^{~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~\left( \text{Magnesium boride} \right)} \\\end{array}\]
\[\begin{align}& 2Mg~+\text{ }Si\to M{{g}_{2}}Si~;~M{{g}_{2}}Si+{{H}_{3}}P{{O}_{4}}\to \text{Mixture of silanes} \\ &~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~{{~}^{(\text{magnesium silicide})}} \\\end{align}\]
(9) The carbides of both Boron and silicon (B4C and SiC) are very hard and used as abrasive.
(10) Oxides of both are acidic and can be reduced by limited amount of Mg In excess of Mg boride and silicide are formed.
\[{{B}_{2}}{{O}_{3}}+3Mg\to 3MgO+2B~;~Si{{O}_{2}}+2Mg\to 2MgO+Si\]
(11) Both the metals and their oxides are readily soluble in alkalies.
\[2B+6NaOH\to \underset{\text{(borate)}}{\mathop{2N{{a}_{3}}B{{O}_{3}}}}\,+3{{H}_{2}}\uparrow \]
\[Si+2NaOH+{{H}_{2}}O\to \underset{\text{(silicate)}}{\mathop{N{{a}_{2}}Si{{O}_{3}}}}\,+\text{ }2{{H}_{2}}\uparrow \]
\[{{B}_{2}}{{O}_{3}}+6NaOH\to 2N{{a}_{3}}B{{O}_{3}}+3{{H}_{2}}O\]
\[Si{{O}_{2}}+2NaOH\to N{{a}_{2}}Si{{O}_{3}}+{{H}_{2}}O\]
Both borates and silicates have tetrahedral structural units and respectively. Boro silicates are known in which boron replaces silicon in the three dimensional lattice. Boron can however form planar BO3 units.
(12) Acids of both these elements form volatile esters on heating with alcohol in presence of conc. H2SO4.
\[B{{(OH)}_{3}}+3ROH\to B{{(OR)}_{3}}+3{{H}_{2}}O\]
\[Si{{(OH)}_{4}}+4ROH\to Si{{(OR)}_{4}}+4{{H}_{2}}O\]
Boron and its compounds
Boron is the first member of group –13 (IIIA) of the periodic table. Boron is a non- metal . It has a small size and high ionization energy due to which it can not lose its valence electrons to form ion. Its compounds especially the hydrides and halides are electron deficient and behave as Lewis acid.
(1) Ores of boron
(i) Borax or tincal : Na2 B4O7 . 10H2O
(ii) Kernite or Rasorite : Na2 B4O7 . 4H2O
(iii) Colemanite : Ca2 B6O11 . 5H2O
(iv) Orthoboric acid : H3BO3 (It occurs in the jets of steam called soffioni escaping from ground in the volcanic region of the Tuscany). Boron is present to a very small extent (0.001%) in earth’s crust.
(2) Isolation : Elemental boron in the form of dark brown powder is obtained either by reduction of boric oxide with highly electropositive metals like K, Mg, Al, Na, etc. in the absence of air and boron halides with hydrogen at high temperature eg.
\[{{B}_{2}}{{O}_{3}}+6K\xrightarrow[{}]{\text{Heat}}~2B+3{{K}_{2}}O\]
\[2BC{{l}_{3}}+3{{H}_{2}}\xrightarrow{1270K}2B+\text{ }6HCl.\]
By thermal decomposition of boron triiodide over red hot tungsten filament and boron hydrides for example,
\[2B{{I}_{3}}\xrightarrow{W,heat}2B+3{{I}_{2}};~{{B}_{2}}{{H}_{6}}\xrightarrow{Heat}2B+3{{H}_{2}}\]
(3) Properties : It exists in mainly two allotropic forms i.e. amorphous dark brown powder and crystalline black very hard solid. It occurs in two isotopic forms, i.e., \[_{5}{{B}^{10}}\] (20% abundance) and \[_{5}{{B}^{11}}\] (80% abundance). With air, boron forms \[{{B}_{2}}{{O}_{3}}\] and BN at 973K, with halogens, trihalides \[(B{{X}_{3}})\] are formed, with metals borides are formed. eg.
\[4B+3{{O}_{2}}\xrightarrow{Heat}\underset{\text{Boron trioxide}}{\mathop{2{{B}_{2}}{{O}_{3}}}}\,\]
\[2B+{{N}_{2}}\xrightarrow{Heat}\underset{\text{Boron nitride}}{\mathop{2BN}}\,\]
\[2B+3{{X}_{2}}\xrightarrow{{}}\underset{\text{Boron trihalide}}{\mathop{2B{{X}_{3}}}}\,\]
\[3Mg+2B\xrightarrow{Heat}\underset{\text{Magnesium boride}}{\mathop{M{{g}_{3}}{{B}_{2}}}}\,\]
Water, steam and HCl have no action on B. oxidising acids \[(HN{{O}_{3}}\], \[{{H}_{2}}S{{O}_{4}})\] convert boron to \[{{H}_{3}}B{{O}_{3}}\].
\[B+3HN{{O}_{3}}\xrightarrow{{}}{{H}_{3}}B{{O}_{3}}+3N{{O}_{2}}\]
\[2B+3{{H}_{2}}S{{O}_{4}}\xrightarrow{{}}2{{H}_{3}}B{{O}_{3}}+3S{{O}_{2}}\]
Fused alkalies (NaOH, KOH) dissolve boron forming borates, liberating hydrogen.
\[2B+6KOH\xrightarrow{Fused}2{{K}_{3}}B{{O}_{3}}+3{{H}_{2}}\]
(4) Uses of Boron : Boron is used in atomic reactors as protective shields and control rods, as a semiconductors for making electronic devices in steel industry for increasing the hardness of steel and in making light composite materials for air crafts.
(5) Compounds of Boron
(i) Boron Hydrides
Boron forms hydrides of the types \[{{B}_{n}}{{H}_{n+4}}\] and \[{{B}_{n}}{{H}_{n+6}}\] called boranes. Diborane is the simplest boron hydride which is a dimer of \[B{{H}_{3}}\].
Preparation
(a) \[8B{{F}_{3}}+6LiH\xrightarrow{450K}{{B}_{2}}{{H}_{6}}+6LiB{{F}_{4}}\]
(b) \[4BC{{l}_{3}}+LiAl{{H}_{4}}\xrightarrow{{}}2{{B}_{2}}{{H}_{6}}+3AlC{{l}_{3}}+3LiCl\]
(c) In the laboratory, it is prepared by the oxidation of sod. Borohydride with \[{{I}_{2}}\].
\[2NaB{{H}_{4}}+{{I}_{2}}\xrightarrow{\text{Polyether}}{{B}_{2}}{{H}_{6}}+2NaI+{{H}_{2}}\]
Properties : (a) Since Boron in boranes never complete its octet of electrons hence all boranes are called as electron-deficient compounds or Lewis acids.
(b) All boranes catch fire in the presence of oxygen to liberated a lot of heat energy. Thus, they can also be used as high energy fuels.
\[{{B}_{2}}{{H}_{6}}+3{{O}_{2}}\xrightarrow{{}}2{{B}_{2}}{{O}_{3}}+3{{H}_{2}}O;\,\Delta H=-1976KJ/mole\]
(c) Boranes are readily hydrolysed by water.
\[{{B}_{2}}{{H}_{6}}+6{{H}_{2}}O\xrightarrow{{}}2{{H}_{3}}B{{O}_{3}}+6{{H}_{2}}\]
(d) With carbon monoxide
\[{{B}_{2}}{{H}_{6}}+2CO\xrightarrow{{}}{{(B{{H}_{3}}\leftarrow CO)}_{2}}\]
(e) Boranes are used for formation of hydroborates or borohydrides such as \[LiB{{H}_{4}}\] or \[NaB{{H}_{4}}\], which are extensively used as reducing agents in organic synthesis.
\[2LiH+{{B}_{2}}{{H}_{6}}\xrightarrow{\text{Diethyl ether }}2L{{i}^{+}}{{[B{{H}_{4}}]}^{-}}\]
\[2NaH+{{B}_{2}}{{H}_{6}}\xrightarrow{\text{Diethyl ether}}2N{{a}^{+}}{{[B{{H}_{4}}]}^{-}}\]
Structure of diborane : \[{{B}_{2}}{{H}_{6}}\] has a three centre electon pair bond also called a banana shape bond.
(a) \[B-{{H}_{t}}\] : It is a normal covalent bond (two centre electron pair bond i.e., \[2c-2e).\]
(b) \[B-{{H}_{b}}\]: This is a bond between three atoms, \[B-{{H}_{b}}-B,\] (three centre electron pair bond i.e., \[3c-2e).\]
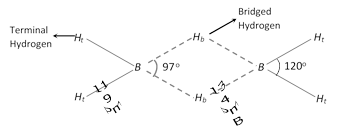
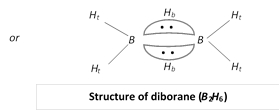
The other boron hydrides are etc.
(ii) Boron Halides
Boron reacts with halogens on strong heating to form boron halides .
\[2B+3{{X}_{2}}\xrightarrow{Heat}2B{{X}_{3}}(X=F,Cl,Br,I)\]
\[B{{F}_{3}}\] and \[BC{{l}_{3}}\] are gases, \[BB{{r}_{3}}\] is a volatile liquid while \[B{{I}_{3}}\] is a solid.
In these halides, the central boron atom has three shared pairs of electrons with the halogen atoms. Therefore, these have two electrons less than the octet and are electron deficient compounds. They acts as Lewis acids.
\[\underset{\text{Lewis acid}}{\mathop{F-\underset{F}{\overset{F}{\mathop{\underset{|}{\overset{|}{\mathop{B}}}\,}}}\,\,\,\,+}}\,\,\,\,\underset{\text{Lewis base}\,\,\,}{\mathop{\underset{\,\,\,H}{\overset{\,\,H}{\mathop{:\underset{|}{\overset{|}{\mathop{N}}}\,}}}\,-H}}\,\]\[\xrightarrow{{}}F-\underset{F}{\overset{F}{\mathop{\underset{|}{\overset{|}{\mathop{B}}}\,}}}\,\xleftarrow{{}}\underset{H}{\overset{H}{\mathop{\underset{|}{\overset{|}{\mathop{N}}}\,}}}\,-H\]
The relative acidic strength of boron trihalides decreases as : \[B{{I}_{3}}>BB{{r}_{3}}>BC{{l}_{3}}>B{{F}_{3}}\].
(iii) Borax (\[N{{a}_{2}}{{B}_{4}}{{O}_{7}}\].\[10{{H}_{2}}O\])
It occurs naturally as tincal (Suhaga) which contains about 50% borax in certain land, lakes. It is also obtained from the mineral colemanite by boiling it with a solution of \[N{{a}_{2}}C{{O}_{3}}\].
\[\underset{\text{Colemanite}}{\mathop{C{{a}_{2}}{{B}_{6}}{{O}_{11}}}}\,+2N{{a}_{2}}C{{O}_{3}}\xrightarrow{{}}\underset{\text{Borax}}{\mathop{N{{a}_{2}}{{B}_{4}}{{O}_{7}}}}\,+2CaC{{O}_{3}}+2NaB{{O}_{2}}\]
Properties : (a) Its aqueous solution is alkaline due to hydrolysis,
\[N{{a}_{2}}\]\[{{B}_{4}}{{O}_{7}}\]+ \[7{{H}_{2}}O\]\[\xrightarrow{{}}\] 2NaOH+4\[H_{3}^{{}}B{{O}_{3}}\].
(b) On heating borax loses its water of crystallization and swells up to form a fluffy mass. On further heating, it melts to give a clear liquid which solidifies to a transparent glassy bead consisting of sodium metaborate (\[NaB{{O}_{2}}\]) and boric anhydride (\[{{B}_{2}}{{O}_{3}}\]),
\[N{{a}_{2}}B{}_{4}{{O}_{7}}.10{{H}_{2}}O\underset{-10{{H}_{2}}O}{\overset{{}}{\mathop{\xrightarrow{\Delta }}}}\,N{{a}_{2}}B{}_{4}{{O}_{7}}\xrightarrow{\Delta }\underset{\text{Borax bead}}{\mathop{2NaB{{O}_{2}}}}\,+B{}_{2}{{O}_{3}}\]
Borax bead is used for the detection of coloured basic radicals under the name borax bead test.
Borax bead test : Borax bead is a mixture of \[NaB{{O}_{2}}\] and \[{{B}_{2}}{{O}_{3}}\]. \[{{B}_{2}}{{O}_{3}}\] on heating combines readily with a number of coloured transition metal oxides such as Co, Ni, Cr, Cu, Mn, etc. to form the corresponding metaborates which possess characteristic colours,
\[CoS{{O}_{4}}\xrightarrow{\Delta }CoO+S{{O}_{3}}\]; \[CoO+{{B}_{2}}{{O}_{3}}\xrightarrow{{}}\underset{\text{(Blue)}}{\mathop{\underset{\text{Cobalt meta borate}}{\mathop{Co{{(B{{O}_{2}})}_{2}}}}\,}}\,\]
Colours of some important metaborates are : Cupric metaborate, Cu\[{{(BO{}_{2})}_{2}}\]is dark blue, chromium metaborate, Cr\[{{(B{{O}_{2}})}_{2}}\] is green, nickel metaborate, \[Ni{{(B{{O}_{2}})}_{2}}\] is brown and manganese metaborate, \[Mn{{(B{{O}_{2}})}_{2}}\] is pink violet.
(c) When heated with \[{{C}_{2}}{{H}_{5}}OH\]and conc. \[{{H}_{2}}S{{O}_{4}}\] it gives volatile vapours of triethyl borate which burns with a green edged flame.
\[N{{a}_{2}}{{B}_{4}}{{O}_{7}}+{{H}_{2}}S{{O}_{4}}+5{{H}_{2}}O\xrightarrow{{}}N{{a}_{2}}S{{O}_{4}}+4{{H}_{3}}B{{O}_{3}}\]
\[{{H}_{3}}B{{O}_{3}}+3{{C}_{2}}{{H}_{5}}OH\xrightarrow{{}}\underset{\text{Triethyl borate}}{\mathop{B{{(O{{C}_{2}}{{H}_{5}})}_{3}}}}\,+3{{H}_{2}}O\]
This reaction is used as a test for borate radical in qualitative analysis.
Uses : (a) In making optical and hard glasses. (b) In the laboratory for borax bead test. (c) In softening of water. (d) In the preparation of medicinal soaps due to its antiseptic character.
(iv) Boric acid or orthoboric acid (\[{{H}_{3}}B{{O}_{3}})\]
It is obtained from borax by treating with dil. HCl or dil. \[{{H}_{2}}S{{O}_{4}}\],
\[N{{a}_{2}}B{}_{4}{{O}_{7}}+2HCl+5{{H}_{2}}O\xrightarrow{{}}2NaCl+4{{H}_{3}}B{{O}_{3}}\]
It can also be obtained from the mineral colemanite by passing \[S{{O}_{2}}\] through a mixture of powdered mineral in boiling water,
\[C{{a}_{2}}{{B}_{6}}\]\[{{O}_{11}}\]+ \[4S{{O}_{2}}+11{{H}_{2}}O\]\[\xrightarrow{{}}2Ca{{(HS{{O}_{3}})}_{2}}+6{{H}_{3}}B{{O}_{3}}\]
Properties : (a) It is a very weak monobasic acid, does not act as a proton doner but behaves as a Lewis acid i.e. it accepts a pair of electrons from \[O{{H}^{-}}\]ion of \[{{H}_{2}}O\],
\[{{H}_{3}}B{{O}_{3}}+{{H}_{2}}O\xrightarrow{{}}{{[B{{(OH)}_{4}}]}^{-}}+{{H}^{+}}\]
It acts as a strong acid in presence of polyhydroxy compounds such as glycerol, mannitol etc. and can be titrated against strong alkali .
(b) With NaOH it forms, sodium metaborate,
\[{{H}_{3}}B{{O}_{3}}+NaOH\xrightarrow{{}}NaB{{O}_{2}}+2{{H}_{2}}O\]
(c) With \[{{C}_{2}}{{H}_{5}}OH\] and conc. \[{{H}_{2}}S{{O}_{4}}\], it gives triethyl borate
\[{{H}_{3}}B{{O}_{3}}\]+3\[{{C}_{2}}{{H}_{5}}OH\]\[\xrightarrow{Conc.\,{{H}_{2}}S{{O}_{4}}}\]B(O\[{{C}_{2}}{{H}_{5}}{{)}_{3}}\]+ 3\[{{H}_{2}}O\]
(d) Action of heat : The complete action of heat on boric acid may be written as,
\[\underset{\text{Boric acid}}{\mathop{{{H}_{3}}B{{O}_{3}}}}\,\xrightarrow{373K}\underset{\text{Metaboric acid}}{\mathop{HB{{O}_{2}}}}\,\xrightarrow{433\,K}\underset{\text{Tetra boric acid}}{\mathop{{{H}_{2}}{{B}_{4}}{{O}_{7}}}}\,\xrightarrow{\text{Red}\,\text{hot}}\underset{\text{Boron oxide}}{\mathop{{{B}_{2}}{{O}_{3}}}}\,\]
Structure : In boric acid, planar \[BO_{3}^{-3}\] units are joined by hydrogen bonds to give a layer structure.
Uses : (a) As a food preservative. (b) As a mild antiseptic for eye wash under the name boric lotion. (c) For the preparation of glazes and enamels in pottery.
(v) Borazine or Borasole or Triborine triamine (\[{{B}_{3}}{{N}_{3}}{{H}_{6}})\]
It is a compound of B, N and H. It is a colourless liquid and is also called inorganic benzene.
2\[{{B}_{2}}{{H}_{6}}+6N{{H}_{3}}\xrightarrow{{{180}^{o}}C}2{{B}_{3}}{{N}_{3}}{{H}_{6}}+12{{H}_{2}}\].
It has a six membered ring of alternating B and N atoms, each is further linked to a H-atom.
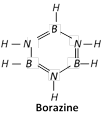
(vi) Boron nitride (BN)
It is prepared by treating \[BC{{l}_{3}}\] with an excess of NH3 and pyrolysing the resulting mixture in an atmosphere of NH3 at 750°C,
\[BC{{l}_{3}}\]+\[N{{H}_{3}}\]\[\xrightarrow{{}}\][\[{{H}_{3}}N\xrightarrow{{}}BC{{l}_{3}}]\]\[\underset{\text{Excess}\,N{{H}_{3}}}{\mathop{\xrightarrow{{{750}^{o}}C}}}\,\]BN + 3HCl .
It is a colourless, good insulator, diamagnetic and almost unreactive solid
Aluminium and its compounds
(1) Ores of Aluminium : Bauxite \[(A{{l}_{2}}{{O}_{3}}.2{{H}_{2}}O),\,\]Cryolite \[(N{{a}_{3}}Al{{F}_{6}},\,\]Felspar \[(KAlS{{i}_{3}}{{O}_{8}}),\] Kaolinite \[(A{{l}_{2}}{{O}_{3}}.\,2Si{{O}_{2}}.2{{H}_{2}}O)\], Mica \[({{K}_{2}}O.3A{{l}_{2}}{{O}_{3}}.\,6Si{{O}_{2}}.2{{H}_{2}}O),\] Corundum \[(A{{l}_{2}}{{O}_{3}})\], Diaspore \[(A{{l}_{2}}{{O}_{3}}.{{H}_{2}}O),\] Alunite or alum stone \[[{{K}_{2}}S{{O}_{4}}.\,A{{l}_{2}}{{(S{{O}_{4}})}_{3}}.\,\] \[4Al{{(OH)}_{3}}].\]
(2) Extraction : Aluminium is obtained by the electrolysis of the oxide (alumina) dissolved in fused cryolite. This involves following steps,
Purification of ore
(i) Baeyer's process
\[\underset{\text{(red)}}{\mathop{\underset{\text{Finely}\ \text{powdered}}{\mathop{\text{Bauxite}}}\,}}\,\underset{FeO\to F{{e}_{2}}{{O}_{3}}}{\mathop{\xrightarrow{\text{Roasted}}}}\,\text{Roasted}\ \text{ore}\ \underset{\text{filtered, F}{{\text{e}}_{\text{2}}}{{O}_{3}}\ \text{as}\,\text{residue}}{\mathop{\underset{\text{High pressure (15}{{\text{0}}^{\text{o}}}\text{C, 80 atm)}}{\mathop{\xrightarrow{+\ \text{Caustic soda solution}}}}\,}}\,\]
\[\ \underset{\text{(Sod}\text{.}\ \text{Aluminate)}}{\mathop{Filtrate}}\,\underset{C{{O}_{2}}}{\mathop{\xrightarrow{Filtered}}}\,Pure\ A{{l}_{2}}{{O}_{3}}\xrightarrow{Heat}Al{{(OH)}_{3}}\]
(ii) Hall's process
\[\underset{\text{(red)}}{\mathop{\underset{\text{(Finely}\ \text{powdered)}}{\mathop{\text{Bauxite}}}\,}}\,\underset{\text{water}\text{. Residue F}{{\text{e}}_{\text{2}}}{{O}_{3}}}{\mathop{\underset{\text{Fused, extracted with}}{\mathop{\xrightarrow{\ \ +\text{N}{{\text{a}}_{\text{2}}}C{{O}_{3}}\ \ \ }}}\,}}\,\text{Solution}\ \underset{\text{Filtrate (N}{{\text{a}}_{\text{2}}}\text{C}{{\text{O}}_{\text{3}}}\text{) }}{\mathop{\underset{\text{50-6}{{\text{0}}^{\text{o}}}\text{C and filtered}\text{.}}{\mathop{\xrightarrow{\ \ \ \ C{{O}_{2\ }}\ \ \ \ \ \ }}}\,}}\,\]
\[\text{Precipitate }Al{{(OH)}_{\text{3}}}\underset{{}}{\mathop{\xrightarrow{Heat}}}\,Pure\ A{{l}_{2}}{{O}_{3}}\]
(iii) Serpek's process
\[\underset{\text{(white)}}{\mathop{\underset{\text{(Finely}\ \text{powdered)}}{\mathop{\text{Bauxite}}}\,}}\,\underset{\text{180}{{\text{0}}^{\text{o}}}\text{C}}{\mathop{\underset{\text{Heated to}}{\mathop{\xrightarrow{\ \ +Coke+{{\text{N}}_{\text{2}}}\ \ }}}\,}}\,\underset{\text{Si which volatalises}}{\mathop{\text{Silica reduced to}}}\,\text{ }\ +\ \underset{\text{aluminium nitride }}{\mathop{\text{Alumina form}}}\,AIN\]\[\xrightarrow{Hydrolysis}\ Pure\ A{{l}_{2}}{{O}_{3}}\underset{{}}{\mathop{\xrightarrow{Heated}}}\,Al{{(OH)}_{3}}\]
(iv) Hall and Heroult process : It is used for extraction of aluminium. In this process a fused mixture of alumina (20%), cryolite (60%) and fluorspar (20%) is electrolysed using carbon electrodes. Whereas cryolite makes \[A{{l}_{2}}{{O}_{3}}\] conducting fluorspar decreases the melting point of alumina.
Aluminium is refined by Hoope's electrolytic process.
(3) Compounds of Aluminium
(i) Aluminium oxide or Alumina \[(A{{l}_{2}}{{O}_{3}})\] : It occurs in nature as colourless corundum and several coloured minerals like ruby (red), topaz (yellow), Sapphire (blue), amethyst (violet) and emerald (green). These minerals are used as precious stones (gems).
(ii) Aluminium chloride \[(A{{l}_{2}}C{{l}_{6}})\] : It is prepared by passing dry chlorine over aluminium powder.
\[A{{l}_{2}}{{O}_{3}}+3C+3C{{l}_{2}}\to \underset{\text{(anhydrous)}}{\mathop{2AlC{{l}_{3}}}}\,+3CO(g)\]
It exists as dimer \[A{{l}_{2}}C{{l}_{6}},\,\] in inert organic solvents and in vapour state. It sublimes at \[100{{\,}^{o}}C\] under vacuum. Dimeric structure disappears when \[AlC{{l}_{3}}\] is dissolved in water. It is hygroscopic in nature and absorbs moisture when exposed to air.
(iii) Thermite : A mixture of aluminium powder and \[F{{e}_{2}}{{O}_{3}}\] in the ratio 1:3. It is used for welding of iron. The reaction between \[Al\] and \[F{{e}_{2}}{{O}_{3}}\] is highly exothermic,
\[Al+F{{e}_{2}}{{O}_{3}}\to A{{l}_{2}}{{O}_{3}}+Fe+\]Heat
(iv) Aluminium sulphate [Al2(SO4)3] : It is used for the preparation of alums e.g., potash alum \[A{{l}_{2}}{{(S{{O}_{4}})}_{3}}.\,{{K}_{2}}S{{O}_{4}}\] \[.\,24{{H}_{2}}O\]. It is also used for making fire proof clothes.
(iv) Alums : In general, the term alum is given to double sulphates of the type \[{{M}_{2}}S{{O}_{4}}.{{{M}'}_{2}}\]\[{{(S{{O}_{4}})}_{3}}.24{{H}_{2}}O\] where \[M\] is a univalent cation like \[N{{a}^{+}},\,{{K}^{+}}\] and \[NH_{4}^{+},\,{M}'\] is a trivalent cation like \[A{{l}^{3+}},F{{e}^{3+}}\] and \[C{{r}^{3+}}\].
Some important points to be noted about the alums are
(a) General formula is \[{{M}_{2}}S{{O}_{4}}.{{{M}'}_{2}}{{(S{{O}_{4}})}_{3}}.24{{H}_{2}}O\]
\[M=\]Monovalent metal; \[{M}'=\]Trivalent metal
In alum crystals, 6 water molecules are held by monovalent ion, 6 water molecules are held by trivalent ion, 12 water molecules are held in the crystal structure.
(b) All alums are isomorphous. Aqueous solutions of alums are acidic due to cationic hydrolysis of trivalent cation.
(c) Double sulphates of divalent ions and trivalent ions with 24 water molecules in their crystals are known as Pseudoalums. General formula is \[MS{{O}_{4}}.{{{M}'}_{2}}{{(S{{O}_{4}})}_{3}}.24{{H}_{2}}O\]
\[M=\] Bivalent metal; \[{M}'=\] Trivalent metal
(d) Pseudoalums are not isomorphous with alums.
(e) Feather alum or ‘Hair-salt’ \[A{{l}_{2}}S{{O}_{4}}.18{{H}_{2}}O\] is a native form of aluminium sulphate.
(f) Potash alum is used for tanning of leather, as mordant in dyeing and calico printing, for sizing paper, as a syptic to stop bleeding and purification of water.
Some important alums are
Potash alum \[{{K}_{2}}S{{O}_{4}}.A{{l}_{2}}{{(S{{O}_{4}})}_{3}}.24{{H}_{2}}O\]
Sodium alum \[N{{a}_{2}}S{{O}_{4}}.A{{l}_{2}}{{(S{{O}_{4}})}_{3}}.24{{H}_{2}}O\]
Ammonium alum \[{{(N{{H}_{4}})}_{2}}S{{O}_{4}}.A{{l}_{2}}{{(S{{O}_{4}})}_{3}}.24{{H}_{2}}O\]
Chrome alum \[{{K}_{2}}S{{O}_{4}}.C{{r}_{2}}{{(S{{O}_{4}})}_{3}}.24{{H}_{2}}O\]
You need to login to perform this action.
You will be redirected in
3 sec
