Answer:
Redox reactions are given below
:
(i)
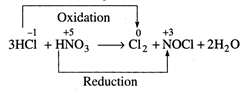 (iii)
(iii)
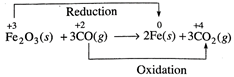 (v)
(v)
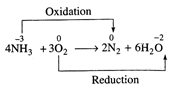
You need to login to perform this action.
You will be redirected in
3 sec
