Answer:
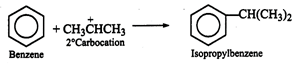
The reaction under consideration
is an example of Friedel-Craft alkylation. The electrophile formed in presence
of
\[AlC{{l}_{3}}\]i.e., \[C{{H}_{3}}-C{{H}_{2}}-\overset{+}{\mathop{C{{H}_{2}}}}\,\]undergo
isomerisation to give more stable \[C{{H}_{3}}-\overset{+}{\mathop{CH}}\,-C{{H}_{3}}\]
ion. The product obtained in the reaction is, therefore, isopropyl benzene or cumene.
\[\underset{n-\text{Propyl}\,chloride}{\mathop{C{{H}_{3}}C{{H}_{2}}C{{H}_{2}}Cl}}\,+AlC{{l}_{3}}\xrightarrow{{}}\underset{{{1}^{{}^\circ
}}Carbocation}{\mathop{C{{H}_{3}}C{{H}_{2}}\overset{+}{\mathop{C{{H}_{2}}}}\,}}\,+AlCl_{4}^{-}\]\[C{{H}_{3}}\underset{(1{}^\circ
)}{\mathop{C{{H}_{2}}}}\,\overset{+}{\mathop{C}}\,{{H}_{2}}\xrightarrow{{}}\underset{(2{}^\circ
)\,more\,\,stable}{\mathop{C{{H}_{3}}}}\,\overset{+}{\mathop{C}}\,HC{{H}_{3}}\]
You need to login to perform this action.
You will be redirected in
3 sec
