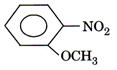
 (ii)
(ii)
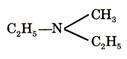
 1
(ii)
1
(ii)
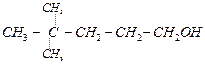 1
(b)
What happens when ethanol is heated with concentrated
1
(b)
What happens when ethanol is heated with concentrated You need to login to perform this action.
You will be redirected in
3 sec
