| Experiment |
time |
Total pressure (atm) |
| I II | 0 100 | 0.5 0.6 |
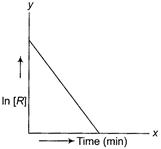 (i) What is the order of the reaction?
(ii) What are the units of rate constant, k for the
reaction? [1+3+1]
(i) What is the order of the reaction?
(ii) What are the units of rate constant, k for the
reaction? [1+3+1]
 |
 |
You need to login to perform this action.
You will be redirected in
3 sec
