-
question_answer1)
What is current?
A)
The flow of matter done
clear
B)
The flow of electrons done
clear
C)
The flow of protons done
clear
D)
The flow of charge done
clear
View Solution play_arrow
-
question_answer2)
In electrolytic solutions, which of the following acts as carrier of charge?
A)
Protons done
clear
B)
Electrons done
clear
C)
Neutrons done
clear
D)
Ions done
clear
View Solution play_arrow
-
question_answer3)
What are insulators?
A)
Materials that conduct electricity done
clear
B)
Materials that do not conduct electricity done
clear
C)
Materials that conduct electricity only at low temperatures done
clear
D)
Materials that conduct electricity at room temperature done
clear
View Solution play_arrow
-
question_answer4)
Which of the following is an insulator?
A)
Wood done
clear
B)
Iron done
clear
C)
Graphite done
clear
D)
Silver done
clear
View Solution play_arrow
-
question_answer5)
Which of the following circuits gives the correct way of connecting an LED to light it up?
A)
B)
C)
D)
View Solution play_arrow
-
question_answer6)
Which of the following energy conversions takes place in a cell?
A)
Electrical energy into chemical energy done
clear
B)
Chemical energy into electrical energy done
clear
C)
Magnetic energy into electrical energy done
clear
D)
Electrical energy into mechanical energy done
clear
View Solution play_arrow
-
question_answer7)
Which of the following are the characteristics of an electrolyte?
A)
It has a positive charge. done
clear
B)
It has a negative charge. done
clear
C)
It conducts charge without dissociating. done
clear
D)
It forms positive and negative ions. done
clear
View Solution play_arrow
-
question_answer8)
Which acid is present in lemon juice that acts as an electrolyte?
A)
Sulphuric acid done
clear
B)
Nitric acid done
clear
C)
Hydrochloric acid done
clear
D)
Citric acid done
clear
View Solution play_arrow
-
question_answer9)
Which of the following is the electrolyte in a dry cell?
A)
Copper sulphate done
clear
B)
Zinc sulphate done
clear
C)
Sulphuric acid done
clear
D)
Ammonium chloride done
clear
View Solution play_arrow
-
question_answer10)
In a dry cell, which of the following acts as the positive terminal?
A)
Carbon rod done
clear
B)
Manganese dioxide done
clear
C)
Manganese dioxide and powdered carbon done
clear
D)
A metal cap on the carbon rod done
clear
View Solution play_arrow
-
question_answer11)
What is the common voltage produced by a dry cell?
A)
1.5 V done
clear
B)
30 V done
clear
C)
60 V done
clear
D)
3 V done
clear
View Solution play_arrow
-
question_answer12)
What happens when electric current is made to flow through a conductor?
A)
Some amount of electrical energy is converted into heat energy. done
clear
B)
Some amount of electrical energy is converted into mechanical energy. done
clear
C)
Some amount of mechanical energy is converted into electrical energy. done
clear
D)
Some amount of heat energy is converted into electrical energy. done
clear
View Solution play_arrow
-
question_answer13)
Nichrome is an alloy. Which of the following metals make up this alloy?
A)
Nickel and chromium done
clear
B)
Nitrogen and chromium done
clear
C)
Nitrogen, chlorine and chromium done
clear
D)
Nickel, chromium and manganese done
clear
View Solution play_arrow
-
question_answer14)
What is the splitting of a compound using electricity called?
A)
Electrolysis done
clear
B)
Electrolyte done
clear
C)
Electrokinesis done
clear
D)
Electrochemistry done
clear
View Solution play_arrow
-
question_answer15)
What is the principle involved in the glowing of an electric bulb?
A)
Magnetic effect of current done
clear
B)
Heating effect of current done
clear
C)
Chemical effect done
clear
D)
Conduction of current done
clear
View Solution play_arrow
-
question_answer16)
Why are LED?s extensively used to replace bulbs?
| (i) Consume less electricity |
| (ii) Have longer life |
| (iii) Have more power |
A)
Only (i) and (ii) done
clear
B)
Only (ii) and (iii) done
clear
C)
Only (i) and (iii) done
clear
D)
(i), (ii) and (iii) done
clear
View Solution play_arrow
-
question_answer17)
Which of the following is the best conductor of electricity?
A)
Tap water done
clear
B)
Distilled water done
clear
C)
Sea water done
clear
D)
Rain water done
clear
View Solution play_arrow
-
question_answer18)
Which of the following effects of current is responsible for the deflection of a compass needle in an electric field?
A)
Heating effect of current done
clear
B)
Magnetic effect of current done
clear
C)
Conducting effect of heat done
clear
D)
Chemical effect of current done
clear
View Solution play_arrow
-
question_answer19)
Which of these is the industrial applications of the chemical effects of electric current?
A)
Electroplating done
clear
B)
Galvanizing done
clear
C)
Anodizing done
clear
D)
All of the above done
clear
View Solution play_arrow
-
question_answer20)
Which of the following liquids is a bad conductor of electricity?
A)
Lemon juice done
clear
B)
Vinegar done
clear
C)
Sea water done
clear
D)
Distilled water done
clear
View Solution play_arrow
-
question_answer21)
Cans used for storing food are electroplated. What is the material used for electroplating?
A)
Chrome onto tin done
clear
B)
Iron onto tin done
clear
C)
Tin onto iron done
clear
D)
Chrome onto iron done
clear
View Solution play_arrow
-
question_answer22)
What is iron coated with in order to protect it from corrosion and rust?
A)
Tin done
clear
B)
Copper done
clear
C)
Zinc done
clear
D)
Mercury done
clear
View Solution play_arrow
-
question_answer23)
Why is zinc better than tin for electro- plating a piece of iron in order to prevent it from rusting?
A)
Zinc is cheaper than tin. done
clear
B)
Tin is toxic. done
clear
C)
Zinc can prevent iron from coming into contact with water and air. done
clear
D)
Rusting is prevented even when the zinc layer is broken. done
clear
View Solution play_arrow
-
question_answer24)
Which of the following statements is correct?
A)
Distilled water is a good conductor of electricity. done
clear
B)
An LED glows even when a weak electric current flows through it. done
clear
C)
Only hydrated salt solutions conduct electricity. done
clear
D)
Zinc plating is done to make the object scratch proof. done
clear
View Solution play_arrow
-
question_answer25)
?X? is a good conductor of electricity. Which of the following could be ?X??
A)
Solid sodium chloride done
clear
B)
Vinegar done
clear
C)
Distilled water done
clear
D)
Liquid oxygen done
clear
View Solution play_arrow
-
question_answer26)
In a cell, by convention, from where does the charge seem to be flowing through?
A)
From positive electrode to negative electrode done
clear
B)
From negative electrode to positive electrode done
clear
C)
Both (A) and (B) done
clear
D)
Cannot be said done
clear
View Solution play_arrow
-
question_answer27)
Why are metals good conductors of electricity?
A)
In metals/outer electrons are strongly bound to the atom. done
clear
B)
In metals, outer electrons are loosely bound to the atom. done
clear
C)
In metals, inner electrons are loosely bound to the atom. done
clear
D)
In metals, protons can detach from the nucleus and conduct electricity. done
clear
View Solution play_arrow
-
question_answer28)
Why are cans used for storing soft drinks or food items usually electroplated with tin?
A)
Tin is less reactive than the metal the can is made of. done
clear
B)
Tin is cheap. done
clear
C)
Tin is strong and shiny in appearance done
clear
D)
Tin is lighter than other metals. done
clear
View Solution play_arrow
-
question_answer29)
A During the electrolysis of \[\text{CuS}{{\text{O}}_{\text{4}}}\]solution, sulphate ions move towards the
A)
copper electrode. done
clear
B)
anode. done
clear
C)
cathode. done
clear
D)
zinc electrode. done
clear
View Solution play_arrow
-
question_answer30)
A bulb does not glow when the probe are hanging in air. Why?
A)
Air absorbs electricity. done
clear
B)
Air is a bad conductor of electricity. done
clear
C)
Electricity is discharged into air. done
clear
D)
Air disperses electricity. done
clear
View Solution play_arrow
-
question_answer31)
Iron vessels are coated with tin to
A)
give better shining. done
clear
B)
increase the strength. done
clear
C)
increase weight. done
clear
D)
prevent rusting. done
clear
View Solution play_arrow
-
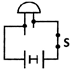
question_answer32)
Observe the given figure.
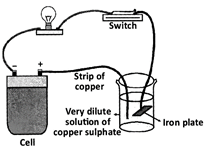
What is the purpose of this experimental setup?
A)
To check the conductivity of copper sulphate solution. done
clear
B)
To get iron electroplated. done
clear
C)
To check the continuity of charge flow in the circuit. done
clear
D)
To make the electric bulb glow. done
clear
View Solution play_arrow
-
question_answer33)
A What is the process of depositing a layer of a desired metal on another material by passing electric current called?
A)
Electrolysis done
clear
B)
Electroplating done
clear
C)
Chromium plating done
clear
D)
Galvanizing done
clear
View Solution play_arrow
-
question_answer34)
A Which of the following statements is NOT correct?
A)
The ions are free to move about in an electrolyte. done
clear
B)
The solution which can form ions is used for electroplating. done
clear
C)
Acids are bad conductors of electricity. done
clear
D)
An electric bulb glows due to the heating effect of current. done
clear
View Solution play_arrow
-
question_answer35)
The charges present on the various objects are shown below in the figure. In which of the following cases does a force of repulsion act between them?
A)
B)
C)
D)
View Solution play_arrow
 done
clear
done
clear
 done
clear
done
clear
 done
clear
done
clear
 done
clear
done
clear
 What is the purpose of this experimental setup?
What is the purpose of this experimental setup? 