States of Matter
Category : 4th Class
Matter is divided into three groups according to the state in which they exist.
![]() Solid
Solid
In solids, the molecules are very close to one another and the force of attraction among the molecules is great.

Arrangement of molecules in solid
Because of this close packing and great attraction among the molecules, solids are hard and has a definite volume and definite shape. For example, ice, book, table, etc.




Books Furniture Animals Plant
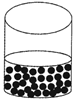
![]() Liquids
Liquids
In liquids, the molecules are not packed very close. They can move about more freely because of less attraction among them.
Arrangement of molecules m liquid
This is the reason for the liquid to flow. So liquid has no definite shape but a definite volume. It takes the shape of the container in which it is put. For example water, milk, cold drink, petrol etc.




Water Milk Petroleum Cold drink
![]() Gas
Gas
In gases, the molecules are far apart from one another. They have lots of freedom to move. Hence gas has neither fixed shape nor fixed volume.
Arrangement of molecules in gas

Water vapour, smoke etc.
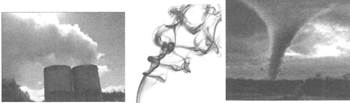

![]() Look at the following figure carefully and choose the correct option for it.
Look at the following figure carefully and choose the correct option for it.

(a) Gas
(b) Liquid
(c) Solid
(d) Both A and C
(e) All the above
Answer: (c)
![]() In gases, the molecules are separated by large spaces and they have freedom to move about. This gives them their character that is
In gases, the molecules are separated by large spaces and they have freedom to move about. This gives them their character that is
(a) No definite shape
(b) No definite volume
(c) Definite volume
(d) (a) and (b)
(e) None of these
Answer: (d)
You need to login to perform this action.
You will be redirected in
3 sec
